Often times engineering students in imperial units systems end up never really learning about lbm and slugs, or it gets glossed over in their statics courses. I think the confusion on this topic arises because physics classes are always going to be taught in metric. 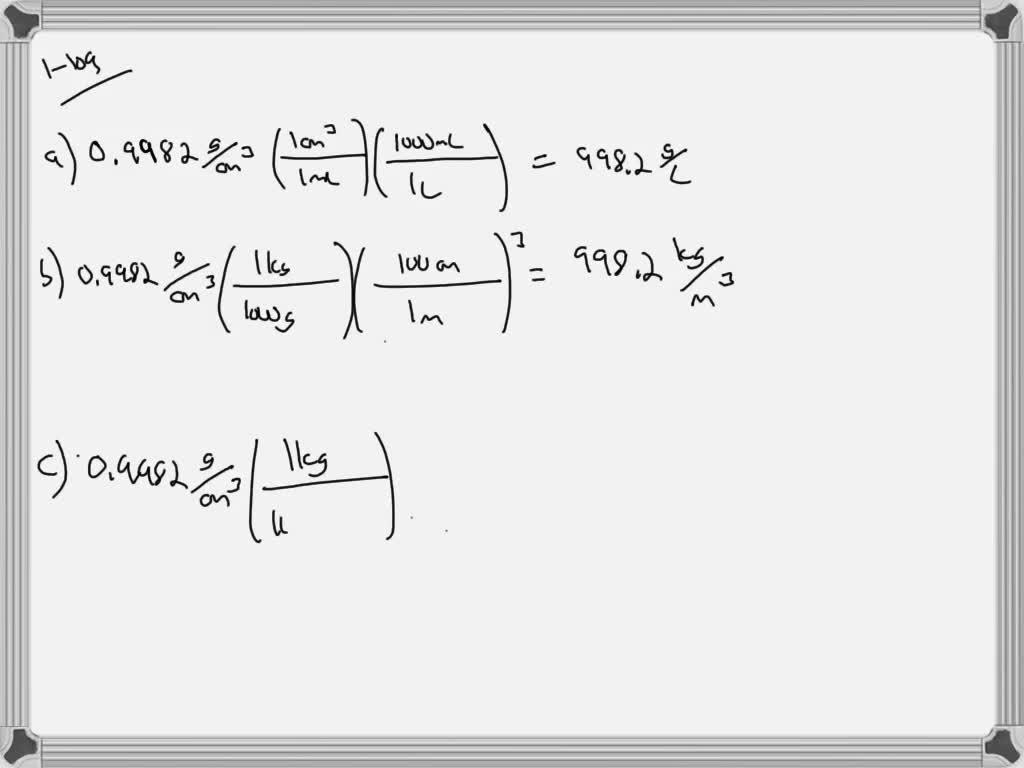
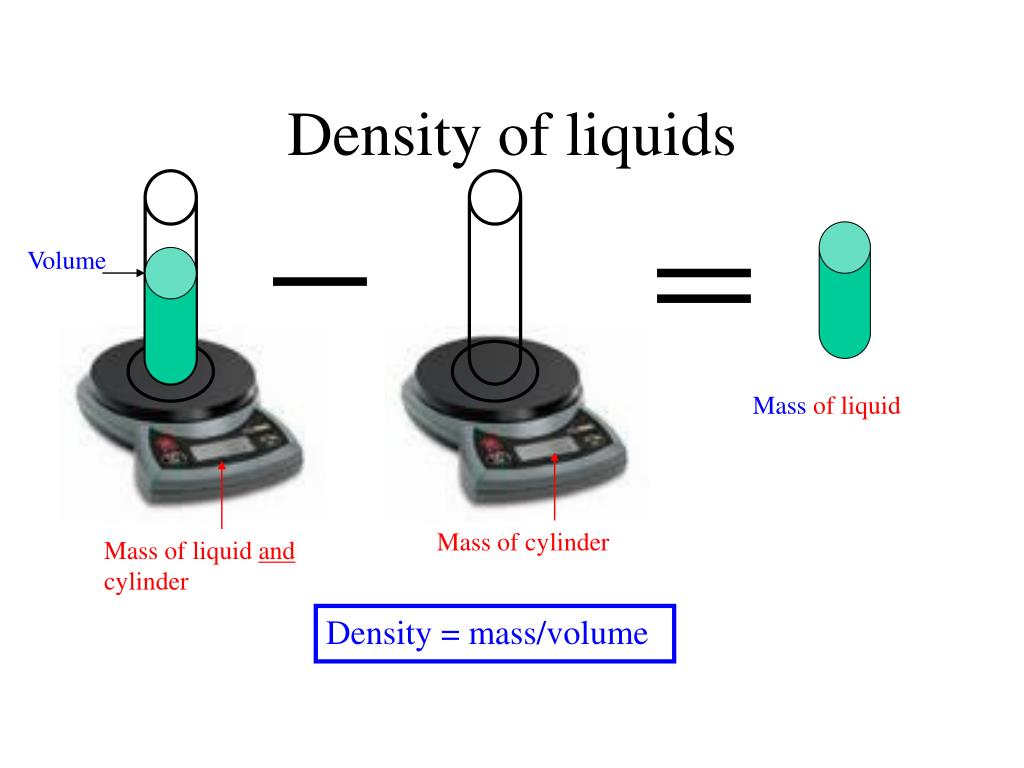
I also don't like that 90% of the time lbs are used in placed of lbf, so you generally end up assuming that pounds = pound force, and that assumption kind of erks me. The magnitude of the units for lbf and lbm being the same for g = 32.2 ft/s2 leads to viewing mass and force as being interchangeable in imperial units, but mass and force are completely different things, same as they are in the metric system. Learn how to find density of a liquid, the difference between ice and water, and the effect of other factors on water density. The exact value is slightly lower and depends on temperature. It depends on temperature and purity, and is about 1 gram per milliliter (g/ml), 1 gram per cubic centimeter (g/cm 3 ), 1000 kg/m 3, or 62 pounds per cubic foot (lb/ft 3 ). The weight of water is approximately 250 tonnes which is a large amount and can affect engineering applications easily. The density of water is the mass of water per unit of volume. The calculator below can be used to calculate the liquid water density at given temperatures. rearranging the density equation m/v to m x v. The density of water is 1.940 sl/ft at 39 F (4 C), and the specific weight in Imperial units is. The density of water is 1 gram/cm 3 since. After you have entered values for two, click on the text representing to third to calculate its value. Specifying any two of the quantities determines the third. Data can be entered into any of the boxes below. The density of water at 10☌ is 999.75 kg/m 3. Density is defined as mass per unit volume. The water reaches its maximum density at a temperature of roughly 4C or 39F, as we all know. density of water at 4 degrees celsius is 998.97 kg/m 3. At-The density of water is maximum at 4 degree Celsius i.e. Therefore, the density of a substance increases with its weight, provided that the volume of the object remains constant. It is the weight of water per unit volume, which is affected by the water's temperature. On the other hand, weight is directly proportional to mass and independent of volume. I personally just wish slugs were always used for mass in place of lbm, because I prefer that clarity of an obvious distinction between mass and force. The volume of water is 10m x 5m x 5m 250m 3. The density of an object is directly proportional to its mass and inversely proportional to its volume. Below is a table of units in which density is commonly expressed, as well as the densities of some common materials. There are exceptions however, such as water's density increasing between 0C and 4C. You've got the right idea, but are confusing lbm for slugs.ĭensity of water in slugs/ft3 = (62.4 lbf) / (32.2 ft/s2) = 1.94 slugs/ft3 Increases in temperature tend to decrease density since the volume will generally increase. Journal of Physical and Chemical Reference Data 31, 387, 2002 DOI 10.1063/1.1461829.Yeah, this is something I dislike in imperial units. 
Pruß: The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use. 
* - NIST Standard Reference Database 69: NIST Chemistry WebBook, Thermophysical Properties of Fluid Systems, Isobaric Properties for Water, 2008. Step 3: According to Table 2, the density of water at room temperature (70☏) is 99.802 of the density of water at 39.2☏ (maximum density). The density of water g/cm 3: 1 g/cm 3: The density of water kg/m 3: 1000 kg/m 3: The density of water lb/ft 3: 62.4 lbs/ft 3: Unit weight of water: app 1 g/cm 3 at 25 degrees Celsius: viscosity of water: 0.01 poise or 0.001 Pa.s (Pascal seconds) at 20 degrees Celsius: the specific gravity of water: 1 at 4 degrees Celsius (no units) a cubic foot. * Temperature according to ITS-90 (International Temperature Scale from 1990). 100 gallons of water weighs 834.54 pounds at maximum density. The following table lists the density D of water as a function of the temperature under isobaric conditions : Density is a physical property - constant at a given temperature and pressure - and may be helpful for identification of substances. The density of water (H 2O) changes depending on temperature and pressure - as with other substances. density of water: 1000 kg/m, 1.938 slugs/ft Unit converter - density On atomic level - particles are packed tighter inside a substance with higher density.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
